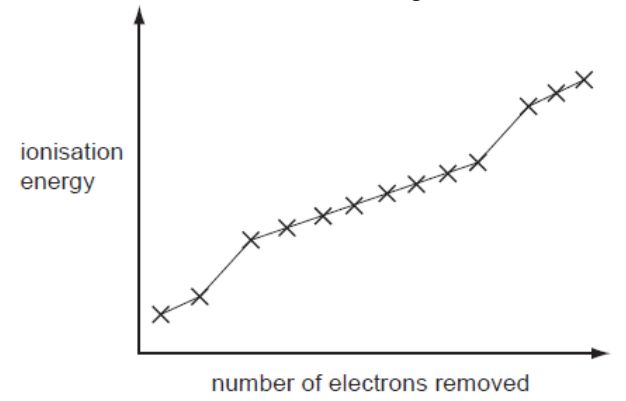
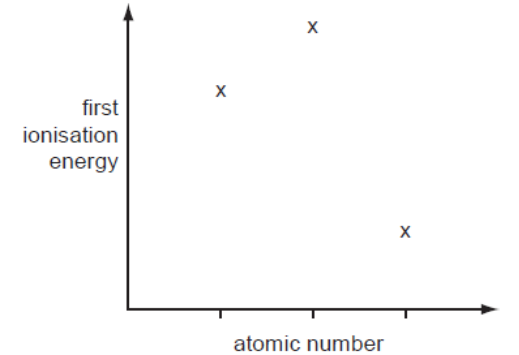
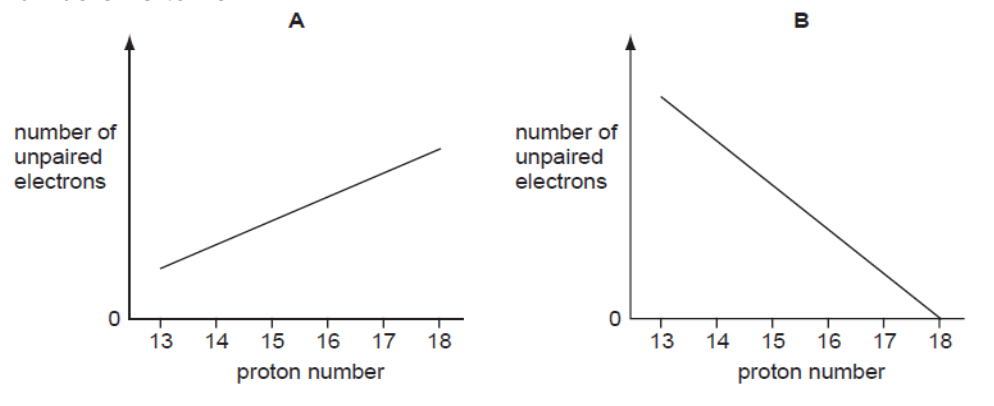
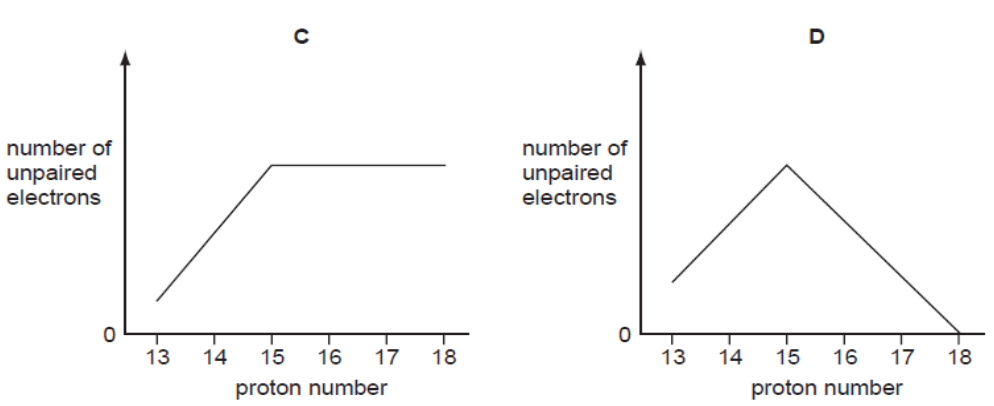
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions
- Multiple Choice Questions
- Structured Questions